 |
What are Steam Traps? |
The duty of a steam trap is to discharge condensate, air and other incondensable gases from a steam system while not permitting the escape of live steam.
The steam trap is an essential part of any steam system. It is the important link between good steam and condensate management, retaining steam within the process for maximum utilisation of heat, but releasing condensate and incondensable gases at the appropriate time.
No steam system is complete without that crucial component 'the steam trap' (or trap). This is the most important link in the condensate loop because it connects steam usage with condensate return.
A steam trap quite literally 'purges' condensate, (as well as air and other incondensable gases), out of the system, allowing steam to reach its destination in as dry a state/condition as possible to perform its task efficiently and economically.
The quantity of condensate a steam trap has to deal with may vary considerably. It may have to discharge condensate at steam temperature (i.e. as soon as it forms in the steam space) or it may be required to discharge below steam temperature, giving up some of its 'sensible heat' in the process.

The pressures at which steam traps can operate may be anywhere from vacuum to well over a hundred bar. To suit these varied conditions there are many different types, each having their own advantages and Disadvantages. Experience shows that steam traps work most efficiently when their characteristics are matched to that of the application. It is imperative that the correct trap is selected to carry out a given function under given conditions. At first sight it may not seem obvious what these conditions are. They may involve variations in operating pressure, heat load or condensate pressure. Steam traps may be subjected to extremes of temperature or even waterhammer. They may need to be resistant to corrosion or dirt. Whatever the conditions, correct steam trap selection is important to system efficiency.
It will become clear that one type of steam trap can not possibly be the correct choice for all applications.
The reason water is so plentiful on the earth is because hydrogen and oxygen are amongst the most abundant elements in the universe. Carbon is another element of significant abundance, and is a key component in all organic matter.
Most mineral substances can exist in the three physical states (solid, liquid and vapour) which are referred to as phases. In the case of H2O, the terms ice, water and steam are used to denote the three phases respectively.
The molecular arrangement of ice, water, and steam is still not fully understood, but it is convenient to consider the molecules as bonded together by electrical charges (referred to as the hydrogen bond).
The degree of excitation of the molecules determines the physical state (or phase) of the substance.
Considerations for steam trap selection
Air venting
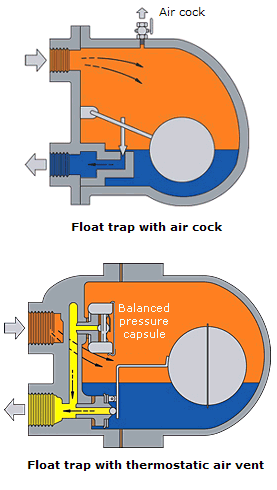
At 'start-up', i.e. the beginning of the process, the heater space is filled with air, which unless displaced, will reduce Heat Transfer and increase the warm-up time. Start-up times increase and plant efficiency falls. It is preferable to purge air as quickly as possible before it has a chance to mix with the incoming steam. Should the air and steam be mixed together they can only be separated by condensing the steam to leave the air, which must then be vented to a safe place. Separate air vents may be required on larger or more awkward steam spaces, but in most cases air in the system is discharged through the steam traps. Here thermostatic traps have a clear advantage over some types of trap since they are fully open at start-up. Float traps with inbuilt thermostatic air vents are especially useful, while many thermodynamic traps are also quite capable of handling moderate amounts of air. However, the small hole in fixed orifice condensate outlets and the bleed hole in inverted bucket traps both vent air slowly. This could increase production times, warm-up times, and corrosion.
Condensate removal
Having vented the air, the trap must then pass the condensate but not the steam. Leakage of steam at this point is inefficient and uneconomical. The steam trap has to allow condensate to pass whilst trapping the steam in the process. If good Heat Transfer is critical to the process, then condensate must be discharged immediately and at steam temperature. Waterlogging is one of the main causes of inefficient steam plant as a result of incorrect steam trap selection.
Plant performance
When the basic requirements of removing air and condensate have been considered, attention may be turned to 'plant performance'. Simply put, unless specifically designed to waterlog, for a Heat Exchanger to operate at its best performance, the steam space must be filled with clean dry steam. The type of steam trap will influence this. For instance, thermostatic traps retain condensate until cooled to below saturation temperature. Should this condensate remain in the steam space, it would reduce the Heat Transfer area and the heater performance. The discharge of condensate at the lowest possible temperature may seem very attractive, but generally most applications require condensate to be removed from the steam space at steam temperature. This needs a steam trap with different operating properties to the thermostatic type, and this usually means either a mechanical or thermodynamic type trap.
Before choosing a particular steam trap it is necessary to consider the needs of the process. This will usually decide the type of trap required. The way in which the process is connected to the steam and condensate system may then decide the type of trap preferred to do the best job under the circumstances. Once chosen, it is necessary to size the steam trap. This will be determined by the system conditions and such process parameters as..
- Maximum steam and condensate pressures.
- Operating steam and condensate pressures.
- Temperatures and flowrates.
- Whether the process is temperature controlled.
Reliability
Experience has shown that 'good steam trapping' is synonymous with reliability, i.e. optimum performance with the minimum of attention.
Causes of unreliability are often associated with the following..
- Corrosion, due to the condition of the condensate. This can be countered by using particular materials of construction, and good feedwater conditioning.
- Waterhammer, often due to a lift after the steam trap, sometimes overlooked at the design stage and often the cause of unnecessary damage to otherwise reliable steam traps.
- Dirt, accumulating from a system where water treatment compound is carried over from the boiler, or where pipe debris is allowed to interfere with trap operation.
The primary task of a steam trap is the proper removal of condensate and air and this requires a clear understanding of how steam traps operate.
Flash steam
An effect caused by passing hot condensate from a high pressure system to a low pressure system is the naturally occurring phenomenon of flash steam. This can confuse the observer regarding the condition of the steam trap.
Consider the enthalpy of freshly formed condensate at steam pressure and temperature (obtainable from steam tables). For example, at a pressure of 7 bar g, condensate will contain 721 kJ/kg at a temperature of 170.5°C. If this condensate is discharged to atmosphere, it can only exist as water at 100°C, containing 419 kJ/kg of enthalpy of saturated water. The surplus enthalpy content of 721 - 419 i.e. 302 kJ/kg, will boil off a proportion of the water, producing a quantity of steam at atmospheric pressure.
How steam traps operate
There are three basic types of steam trap into which all variations fall, all three are classified by International Standard ISO 6704:1982.
Types of steam trap..
- Thermostatic (operated by changes in fluid temperature) - The temperature of saturated steam is determined by its pressure. In the steam space, steam gives up its enthalpy of evaporation (heat), producing condensate at steam temperature. As a result of any further heat loss, the temperature of the condensate will fall. A thermostatic trap will pass condensate when this lower temperature is sensed. As steam reaches the trap, the temperature increases and the trap closes.
- Mechanical (operated by changes in fluid density) - This range of steam traps operates by sensing the difference in density between steam and condensate. These steam traps include 'ball float traps' and 'inverted bucket traps'. In the 'ball float trap', the ball rises in the presence of condensate, opening a Valve which passes the denser condensate. With the 'inverted bucket trap', the inverted bucket floats when steam reaches the trap and rises to shut the Valve. Both are essentially 'mechanical' in their method of operation.
- Thermodynamic (operated by changes in fluid dynamics) - Thermodynamic steam traps rely partly on the formation of flash steam from condensate. This group includes 'thermodynamic', 'disc', 'impulse' and 'labyrinth' steam traps.
Also loosely included in this type are 'fixed orifice traps', which cannot be clearly defined as automatic devices as they are simply a fixed diameter hole set to pass a calculated amount of condensate under one set of conditions.
All rely on the fact that hot condensate, released under dynamic pressure, will flash-off to give a mixture of steam and water.
Reference.. Spirax Sarco
International and european standards relating to steam traps
ISO 6552.. 1980
(BS 6023.. 1981)
Glossary of technical terms for automatic steam traps
ISO 6553.. 1980
CEN 26553.. 1991 (Replaces BS 6024.. 1981)
Marking of automatic steam traps
ISO 6554.. 1980
CEN 26554.. 1991 (Replaces BS 6026.. 1981)
Face-to-face dimensions for flanged automatic steam traps
ISO 6704.. 1982
CEN 26704.. 1991 (Replaces BS 6022.. 1983)
Classification of automatic steam traps
ISO 6948 :1981
CEN 26948.. 1991 (Replaces BS 6025.. 1982)
Production and performance characteristic tests for automatic steam traps
ISO 7841.. 1988
CEN 27841.. 1991 (Replaces BS 6027.. 1990)
Methods for determination of steam loss of automatic steam traps
ISO 7842.. 1988
CEN 27842.. 1991 (Replaces BS 6028.. 1990)
Methods for determination of discharge capacity of automatic steam traps
